Draw The Electron Configuration For A Neutral Atom Of Neon
Draw The Electron Configuration For A Neutral Atom Of Neon - Web in writing the electron configuration for neon the first two electrons will go in the 1s orbital. An atom has a valence shell electron configuration of #ns^1#. Electron configuration through orbit (bohr principle) electron configuration through orbital (aufbau principle) neon (ne) atom electron configuration through orbitals follows different principles. Web now if we take a neutral atom of neon, ne, which is just to the right of fluorine, neon is practically the opposite of fluorine in terms of reactivity despite both being neutral atoms. The alkali metal sodium (atomic number 11) has one more electron than the neon atom.
Sodium (na) has atomic number 11, hence, 11 electrons. First ionisation energy the minimum energy required to remove an electron from a neutral atom in its. Web what is the electron configuration of a neutral atom of sodium (na)? Web how to draw an electron configuration diagram. Web in writing the electron configuration for neon the first two electrons will go in the 1s orbital. A neutral helium atom, with an atomic number of. Web find the atomic number of nitrogen (7) and use this electron configuration calculator to get a complete electron configuration.
Neon Element (Ne 10) of Periodic Table Periodic Table FlashCard
Web in writing the electron configuration for neon the first two electrons will go in the 1s orbital. However, it's easy to determine the configuration of electrons for. We describe an electron configuration with a.
Neon (Ne) Electron Configuration YouTube
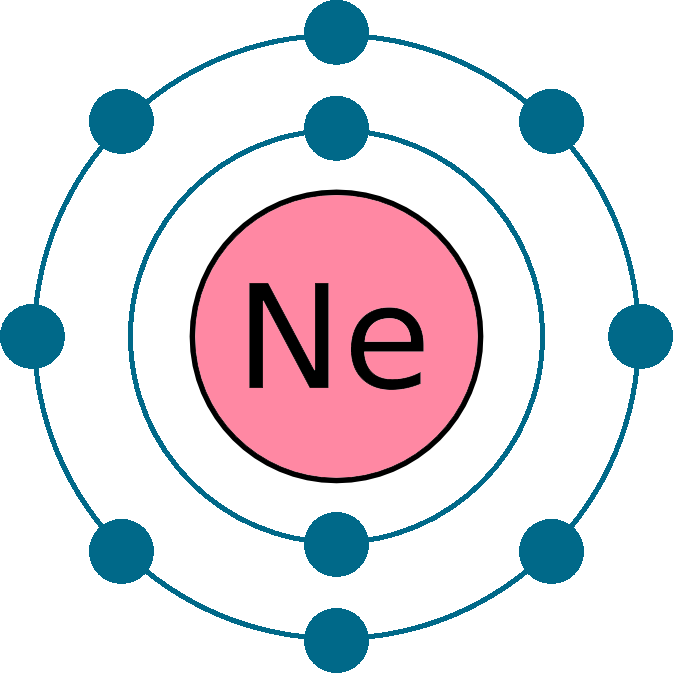
Web how to draw an electron configuration diagram. Electron configuration through orbit (bohr principle) electron configuration through orbital (aufbau principle) neon (ne) atom electron configuration through orbitals follows different principles. This means the first shell.
What is the groundstate electron configuration of a neutral atom of
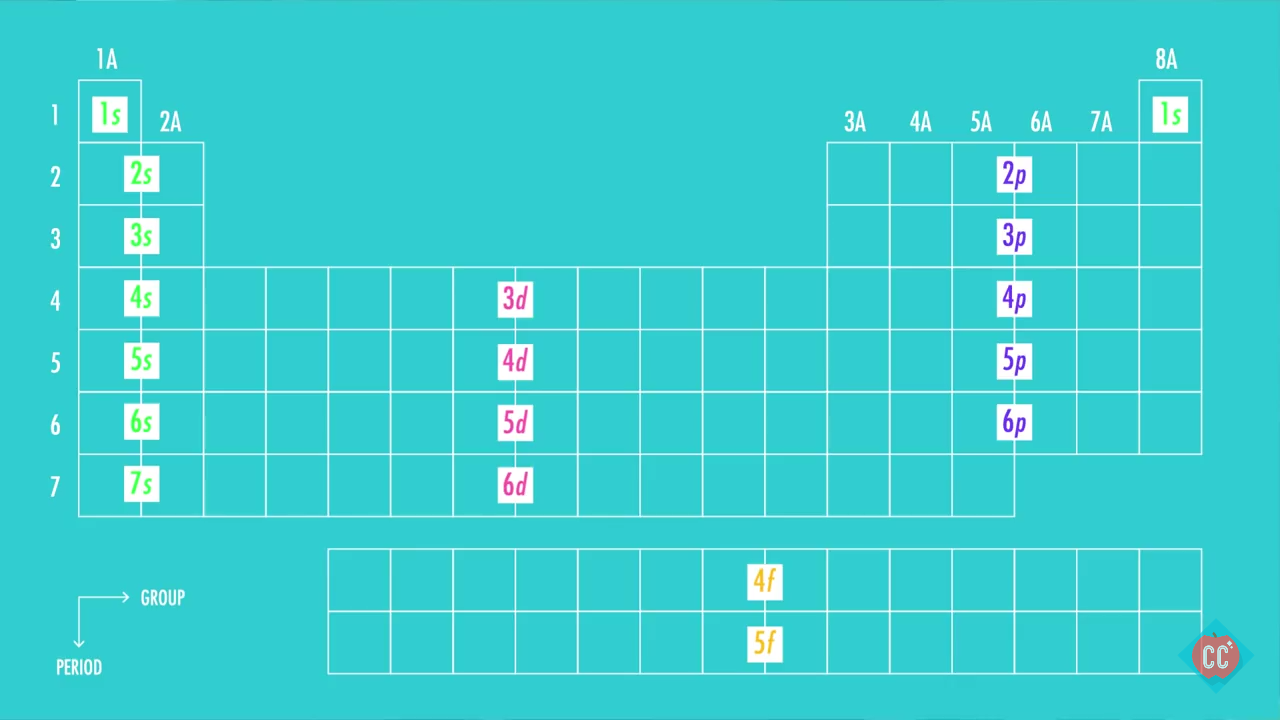
How many core electrons are there? Web to find the electron configuration for an ion, first identify the configuration for the neutral atom. The number of the principal quantum shell, n, the letter that designates.
See the Electron Configuration of Atoms of the Elements Neon Atom
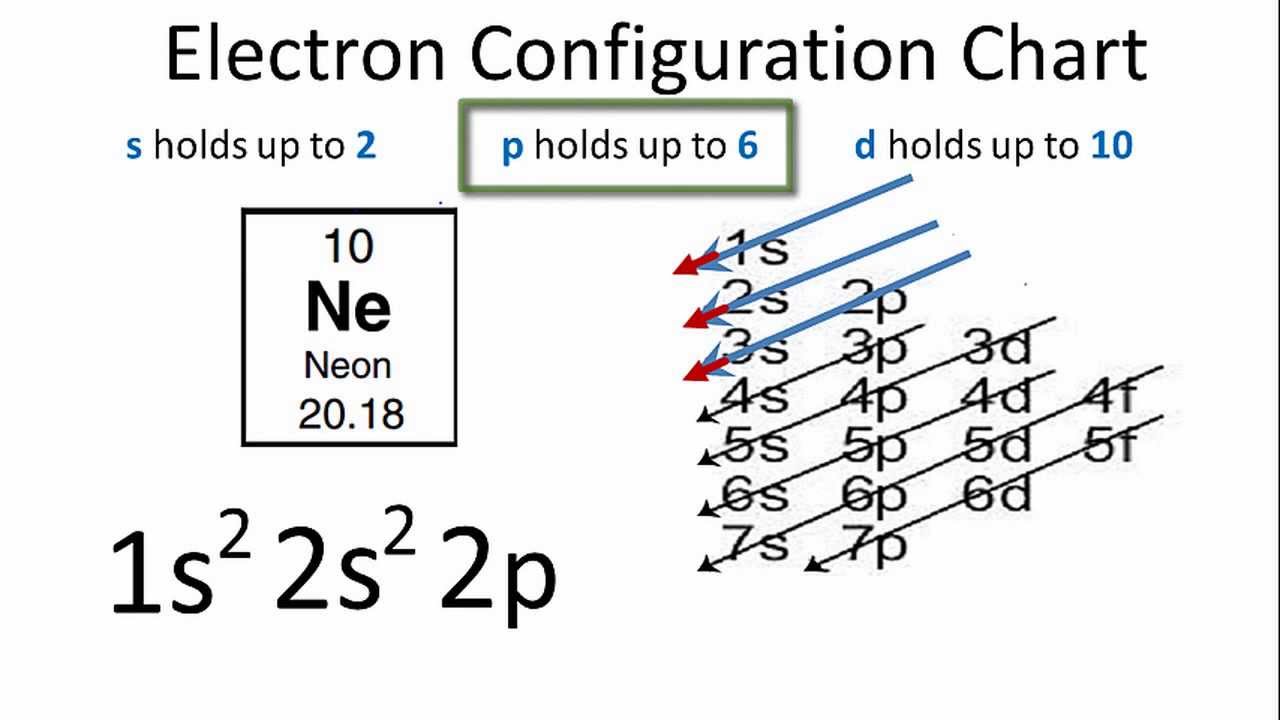
Now, for the electron configuration of neon, the first 2 electrons will go in 1s orbital since s subshell can hold a maximum of 2 electrons. Web electron configurations are a simple way of writing.
Neon Atom Science Notes and Projects
We find the configuration to be 1s22s22p6 which we know is correct because if you add up the superscripts, you'll get the total number of electrons; Web the electron configuration of neon is [ he].
Diagram representation element neon Royalty Free Vector
The electron configurations and orbital diagrams of these four elements are: Web what is the electron configuration of a neutral atom of sodium (na)? Answer the electron configuration for a neutral atom of neon is.
Electron Configuration for Neon (Ne) Full Explanation
Since 1s can only hold two electrons the next 2 electrons for ne go in the 2s orbital. Web electron affinity the energy released when an electron is added to the neutral atom and a.
Atom Diagrams Electron Configurations of the Elements
Draw a small circle and write the symbol in the centre. The atomic number tells you how many electrons to draw in total. Electron configuration can be done in two ways. A neutral helium atom,.
Solved Draw the electron configuration for a neutral atom of
Because the second energy level (2s 2 2p 6. This means the first shell (1s) has 2. Then, add or remove electrons depending on the ion's charge. First ionisation energy the minimum energy required to.
Neon Electron Configuration (Ne) with Orbital Diagram
An atom has a valence shell electron configuration of #ns^1#. Neon has atomic number 10, so a neon atom has 10 protons in its nucleus and therefore 10 electrons. Then, add or remove electrons depending.
Draw The Electron Configuration For A Neutral Atom Of Neon Web an electrically neutral atom has the following electron configuration: However, it's easy to determine the configuration of electrons for. The atomic number tells you how many electrons to draw in total. For example, to find the configuration for the lithium ion (li⁺), start with neutral lithium (1s²2s¹). What is the name of this atom?





:max_bytes(150000):strip_icc()/neonatom-58b602755f9b5860464c7e5a.jpg)

