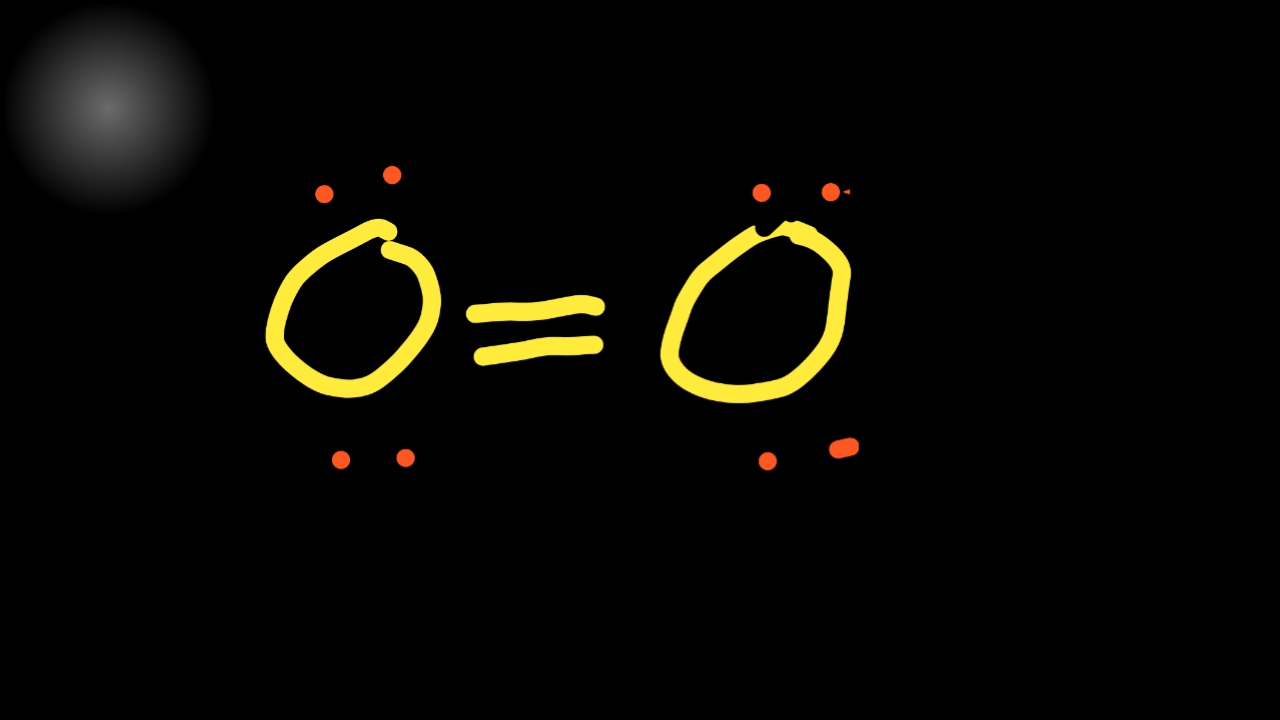Draw The Lewis Structure For A Oxygen O2 Molecule
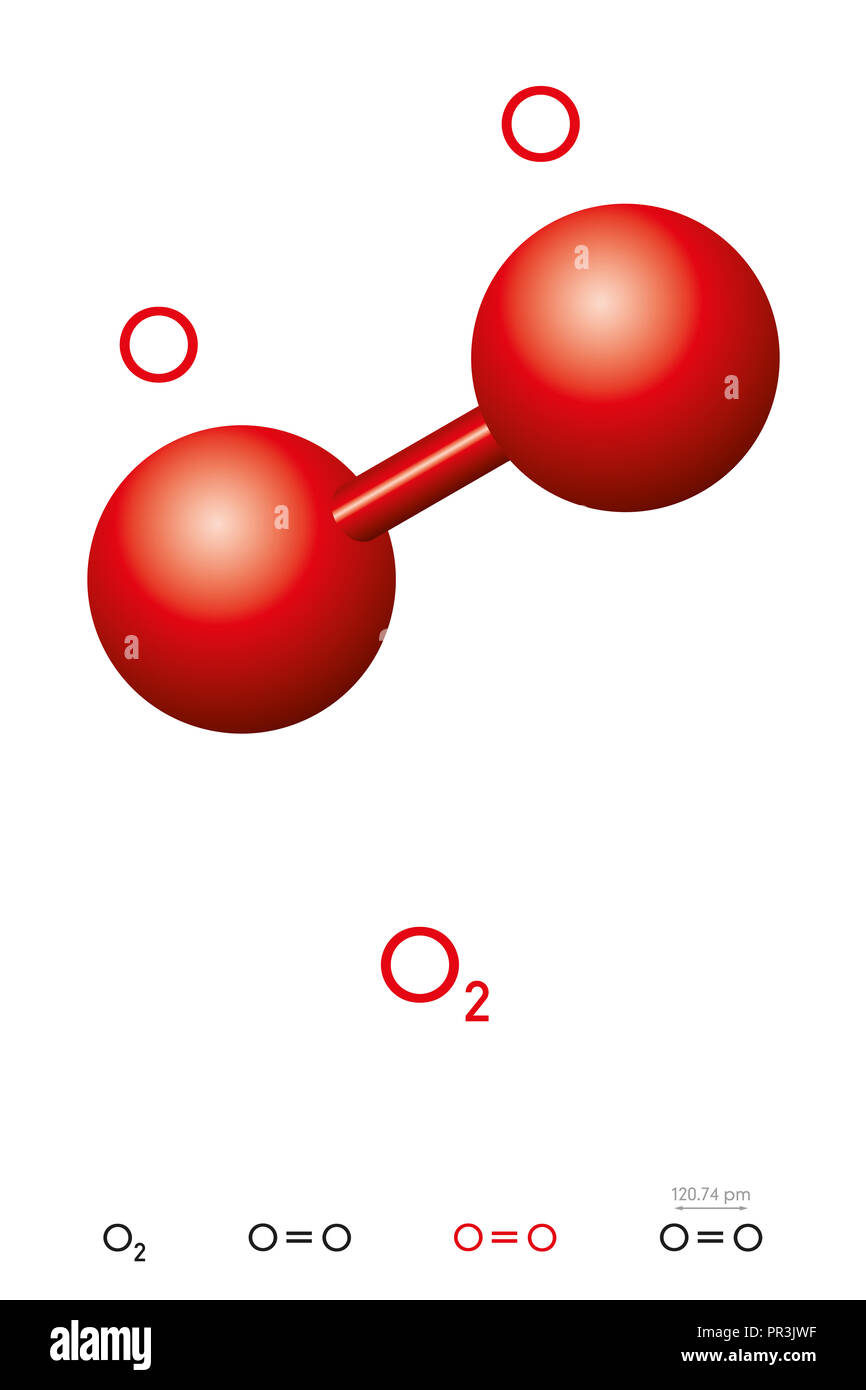
Draw The Lewis Structure For A Oxygen O2 Molecule - In the lewis structure of o 2 molecule, a double bond is located between oxygen atoms and each oxygen atom has two lone pairs in their valence shells. Web to draw the lewis structure of o2, we first need to determine the number of valence electrons for each oxygen atom. Here, the given molecule is o2 (oxygen). Be sure to draw all bonds and lone pairs. Drawing the lewis structure for o 2 ( dioxygen or oxygen gas)
Find the total valence electrons in o2 molecule in order to find the total valence electrons in o2 (oxygen) molecule, first of all you should know the valence electrons present in a single oxygen atom. Note that each atom must contribute one electron to the bond. Steps for writing lewis structures. Each h atom (group 1) has 1 valence electron, and the o atom (group 16) has 6 valence electrons, for a total of 8 valence electrons. Oxygen is in group 16 of the periodic table, so it has 6 valence electrons. Web 6 steps to draw the lewis structure of o2 step #1: In order to draw the lewis structure of o2, first of all you have to find the total number of valence electrons present in the o2 molecule.
O2 oxygen molecule Royalty Free Vector Image VectorStock
The first step is to sketch the lewis structure of the o2 molecule, to add valence electrons around the two oxygen atoms, and the final step is to combine the two oxygen diatomic atoms to.
How to Draw the Lewis Dot Structure for O2 Oxygen gas YouTube
Each h atom (group 1) has 1 valence electron, and the o atom (group 16) has 6 valence electrons, for a total of 8 valence electrons. Web in the lewis structure of o2 structure there.
Oxygen, O2, molecule model and chemical formula. Also dioxygen
Find the total valence electrons in o2 molecule in order to find the total valence electrons in o2 (oxygen) molecule, first of all you should know the valence electrons present in a single oxygen atom..
How to Draw O2 Lewis Structure? 3
In the lewis structure of o 2 molecule, a double bond is located between oxygen atoms and each oxygen atom has two lone pairs in their valence shells. Each h atom (group 1) has 1.
O2 Lewis Structure, Molecular Geometry, and Hybridization Techiescientist
Calculate the total number of valence electrons. We place three lone pairs of electrons around each f atom, accounting for 36 electrons. These four valence electrons form two shared pairs of covalent bonds, providing a.
【2 Step】O2 Lewis StructureLewis Dot Structure for Oxygen(O,O2)Lewis
Web lewis structures, introduction, formal charge, molecular geometry, resonance, polar or nonpolar. Web o 2 lewis structure. Note that each atom must contribute one electron to the bond. Drawing the lewis structure for o 2.
O2 2 Lewis Structure How to Draw the Lewis Structure for O2 2 YouTube
1st attempt part 1 (1 point) draw the lewis structure for o2. Web to draw the lewis structure of o2, we first need to determine the number of valence electrons for each oxygen atom. Web.
O2 Lewis Structure
Drawing the lewis structure for o 2 ( dioxygen or oxygen gas) The lewis diagram of o2 shows two oxygen atoms having twelve dots, of valence electrons. Web how to draw the lewis dot structure.
O2 Lewis Structure, Molecular Geometry, and Hybridization Techiescientist
These four valence electrons form two shared pairs of covalent bonds, providing a stable structure to the oxygen molecule. Web lewis structure of o2. Web 6 steps to draw the lewis structure of o2 step.
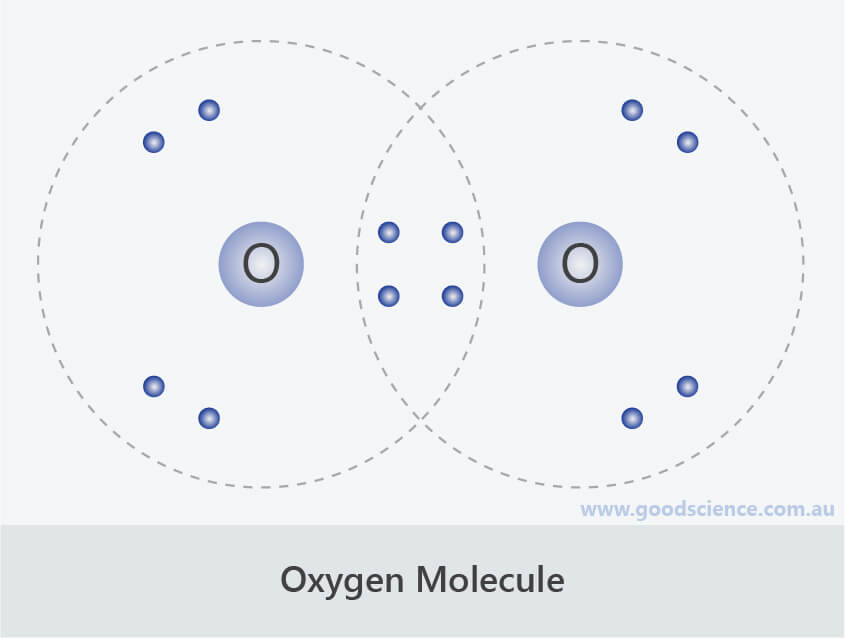
Ionic and Covalent Compounds Good Science
In a water molecule, an oxygen atom forms two bonds, one to each hydrogen atom. These four valence electrons form two shared pairs of covalent bonds, providing a stable structure to the oxygen molecule. Web.
Draw The Lewis Structure For A Oxygen O2 Molecule Here we will take co 2 molecule as an example to explain the procedure step by step:. Two fluorine atoms can form a molecule of f 2 in the same fashion. Total number of valence electrons: Where six are arranged, around each oxygen atom in a way that one side has four valence electrons. Be sure to draw all bonds and lone pairs.